 /phase-changes-56a12ddd3df78cf772682e07.png)
The energy involved in this phase change is called the heat of vaporization or enthalpy of vaporization (ΔHvap). The shift from liquid to gas is named evaporation. 
Melting: The energy required to break intermolecular forces and increase particle motion during a solid-liquid transition is called the heat of fusion or enthalpy of fusion (ΔHfus) while in evaporation, the speed of particle movement continues to increase with increasing temperature. The reversal process is an endothermic process, which means that energy is released from the substance The change from solid to liquid, solid to gas, and liquid to gas is an endothermic process, meaning energy must be added. From liquid to gas, this is called evaporation, and from gas to liquid condensation. A change from solid to liquid is known as melting, and from liquid to solid is known as freezing. The phase change from solid to gas is called sublimation, and from gas to solid deposition. To move from one state of matter to another, we need to add enough energy to overcome the intermolecular forces holding the particles together. The chemical identity of the particles in a liquid determines the nature and strength of the intermolecular attractions. Phase change: The state of matter depends on the intermolecular forces (IMF) and kinetic energy (KE) of the particles - be they molecules, ions, or atoms. The arrangement of the molecules in a matter determines the different properties of solids, liquids, and gases. Gases, also known as vapors, take on the volume and shape of their container it does not have a definite shape and volume. The liquid takes the shape of its container has a certain volume but does not have a certain shape. In the gaseous form, they move freely, in the liquid form, they have a more limited range of motion, and in the solid form, they do not change position but vibrate within a place.įigure 2: Three states of a water molecule. Water molecules move differently depending on their physical state. Three states of matter exist on earth: solid, liquid, and gas. Not being able to see the processes and having few relevances to the real world can make studying it unappealing and make it hard for students to stay motivated. Since matter and phase changes occur at the molecular level, you can’t see or feel them. Let's discuss three reasons why matter and phase changes can prove to be tough, even for an academically sound student. Why Matter And Phase Changes can be tricky Keep reading to know why this topic can be challenging for teachers and their students, five tactics to improve the situation, and learn the benefits of a virtual lab. Solids, liquids, and gases are three states of matter naturally found on earth.ĭepending on its composition, matter can be classified into several categories, as shown in figure 1, either as a mixture or pure substance. Remember, the solid and liquid phases of H 2O can coexist at 0☌.Matter is described as anything that occupies space and has mass. If heat is added to ice at 0☌, some of the solid changes phase to make liquid, which is also at 0☌. In other words, phase changes are isothermal (isothermal means “constant temperature”). 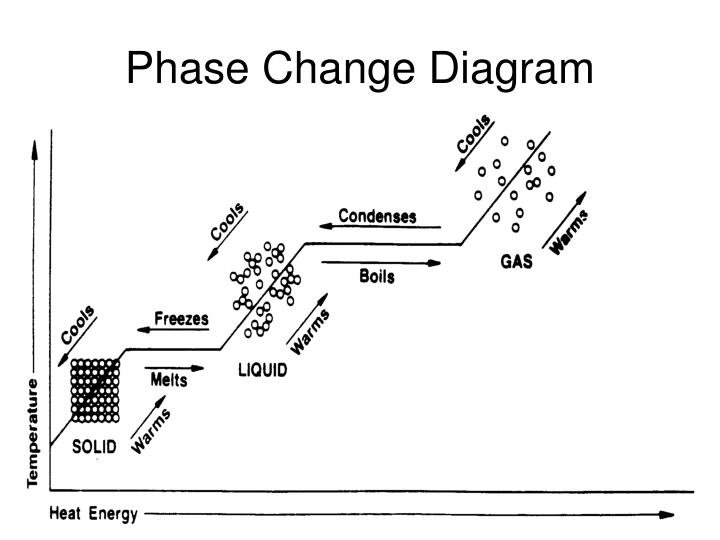
\), the temperature of a substance does not change as the substance goes from one phase to another. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
